A physico-chemo-electrochemically coupled stable interface for high-capacity and durable aqueous zinc metal batteries†
Abstract
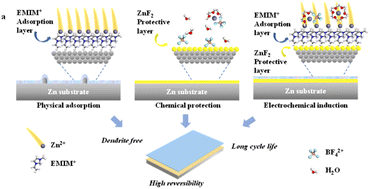
Aqueous zinc metal batteries (AZMBs) as one of the next-generation energy storage technologies have attracted both academic and industrial attention due to their high safety, low cost, and high specific energy, yet are facing interfacial challenges at the electrolyte/anode interface, e.g. uneven physical diffusion of Zn2+ flux, severe chemical corrosion, and disordered electrochemical deposition behavior of the zinc metal anode. To address these issues, herein, we propose a facile and effective electrolyte modulating strategy to construct a physico-chemo-electrochemically coupled stable interface between the aqueous electrolyte and zinc anode. The aqueous electrolyte modified using a tiny amount of water-soluble ionic liquid 1-ethyl-3-methylimidazolium tetrafluoroborate (EMIMBF4) can form a positively charged organic EMIM+ adsorption layer and a protective inorganic Zn2+-conducting layer through the in situ decomposition of BF4− on the zinc anode surface, thus effectively homogenizing the Zn2+ diffusion flux and decreasing the chemical corrosion rate. Moreover, EMIM+ provides an electrostatic shielding layer, leading to the preferential and ordered electrodeposition of Zn2+ along the Zn (002). Benefiting from this physico-chemo-electrochemically multifunctional interfacial layer, the Zn//Zn symmetric cells achieve a considerably extended lifespan of up to 1200 h at 5 mA cm−2 and 5 mA h cm−2, and more than 450 h even at an ultrahigh current density and large deposition capacity (10 mA cm−2 and 10 mA h cm−2). Zn//Cu asymmetric cells also exhibit excellent reversibility with a high coulombic efficiency of 99.75%. Furthermore, the physically, chemically, and electrochemically stabilized interface enables the AZMB with a quinone-based cathode to offer high specific capacity (256.9 mA h g−1) and a decent capacity retention rate (83.9% after 300 cycles).


 Please wait while we load your content...
Please wait while we load your content...