Self-assembly of metal–organosilicate on porous silica substrates for efficient CO2 hydrogenation to methanol†
Abstract
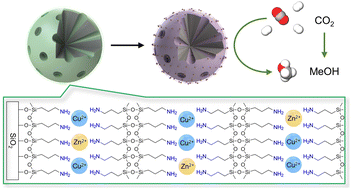
CuZn-based nanocatalysts for the application of MeOH synthesis from the hydrogenation of CO2 have been well-studied in the past few decades. However, many research endeavours have focused on individual aspects of the catalyst design such as the engineering of the support structure alone. In this study, we demonstrate the multiplying effect of an integrated design of both the active phase and support architecture. An active phase consisting of Cu–ZnO nanoparticles embedded inside a microporous silica matrix was derived from a self-assembled copper–organosilicate shell coated on a porous spherical silica support. The surface-silanol-induced self-assembly was carried out via simple one-pot synthesis. The obtained catalyst achieved a specific MeOH yield of 1634 mg MeOH per g Cu per h at 260 °C, 30 barg, as well as long-term stability over a 200 h on-stream operation. These results are attributed to the small Cu–ZnO nanoparticle dimensions with their decent dispersion and blending facilitating the strong metal–support interaction (SMSI) and the spatial confinement effect of the microporous silica matrix. Moreover, the appropriate silica substrate not only promoted self-assembly during the catalyst synthesis but also enhanced the fluid dynamics inside the packed bed. As a result, a higher single-pass CO2 conversion was attained by the porous silica-supported active phase.


 Please wait while we load your content...
Please wait while we load your content...