Nano-scale new Heusler compounds NiRh2Sb and CuRh2Sb: synthesis, characterization, and application as electrocatalysts†
Abstract
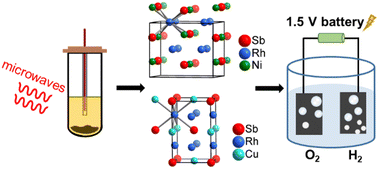
Two Heusler compounds NiRh2Sb and CuRh2Sb were synthesized for the first time and their crystal structures were determined by combined X-ray diffraction and high-resolution electron microscopy. Two synthesis routes were applied: a one-pot polyol reduction of metal cations at 280–290 °C under microwave radiation yielded nanocrystalline particles of 3–6 nm, whereas intergrown microcrystalline samples were obtained by conventional high-temperature synthesis at 700 °C starting from the elements. NiRh2Sb is an orthorhombic Heusler compound that crystallizes in space group Cmcm with lattice parameters a = 6.9188(1) Å, b = 5.7917(1) Å and c = 5.7374(1) Å. CuRh2Sb is dimorphic; depending on the synthesis conditions, it either adopts the cubic space group Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m with a lattice parameter of a = 6.1235(1) Å, or the tetragonal space group I4/mmm with lattice parameters a = 4.1859(1) Å and c = 6.6375(1) Å. The transition between the cubic and tetragonal structures of CuRh2Sb is attributed to a reversible band Jahn–Teller distortion as suggested by density functional theory-based calculations. Electrical resistivities evidence metallic behavior for both compounds in accordance with the computed band structures and the density-of-states. Both compounds are Pauli paramagnetic and show no magnetic ordering between room temperature and 2 K. The electrocatalytic performances of the nano-scale intermetallics were tested with the electrolysis of water, and both were found to greatly reduce the overpotential of both the hydrogen evolution reaction (HER) and the oxygen evolution reaction (OER) with a large electrochemically active surface area.
m with a lattice parameter of a = 6.1235(1) Å, or the tetragonal space group I4/mmm with lattice parameters a = 4.1859(1) Å and c = 6.6375(1) Å. The transition between the cubic and tetragonal structures of CuRh2Sb is attributed to a reversible band Jahn–Teller distortion as suggested by density functional theory-based calculations. Electrical resistivities evidence metallic behavior for both compounds in accordance with the computed band structures and the density-of-states. Both compounds are Pauli paramagnetic and show no magnetic ordering between room temperature and 2 K. The electrocatalytic performances of the nano-scale intermetallics were tested with the electrolysis of water, and both were found to greatly reduce the overpotential of both the hydrogen evolution reaction (HER) and the oxygen evolution reaction (OER) with a large electrochemically active surface area.


 Please wait while we load your content...
Please wait while we load your content...