Photoluminescent supramolecular gels derived from phenylenediamine hydrochlorides†
Abstract
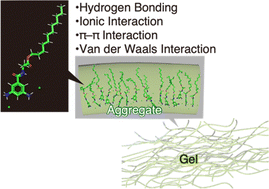
Supramolecular gels are promising as chemosensors, drug delivery systems and oil gelators. The present paper deals with photoluminescent supramolecular gels derived from phenylenediamine hydrochlorides. N-(3,5-Diaminobenzoyl)-L-alanine dodecyl ester dihydrochlorides (1L) underwent gelation in tetrahydrofuran (THF) and CHCl3, but not in C1–C4 alcohols, dimethyl sulfoxide (DMSO) and N,N-dimethylformamide (DMF). Compound 1L emitted blue fluorescence in the sol state and green fluorescence in the gel state. A THF solution of 1L exhibited absorption and emission maxima at wavelengths 94–104 nm and 92–110 nm longer than those in other solvents, such as methanol and ethanol, that cause no gelation of 1L. Particles with hydrodynamic diameters around 13 nm were detected in a THF solution (c = 1.0 mM) of 1L. The gelation of 1L in THF and CHCl3, and the lack of gelation in MeOH were supported by molecular dynamics simulations and dynamic light scattering measurements. N-(3,5-Diaminobenzoyl)-L-alanine dodecyl ester (1L′), an HCl-free analogous compound of 1L, underwent no gelation in THF and CHCl3, indicating the necessity of the ammonium salt structure for gelation. The UV-vis absorption and photoluminescence spectroscopic peaks of 1L red-shifted upon aggregation, which was supported by TD-DFT calculations of monomeric and dimeric models of 1L.


 Please wait while we load your content...
Please wait while we load your content...