Renewable fuel intermediates from furfural over copper-loaded mesoporous aldol condensation catalysts†
Abstract
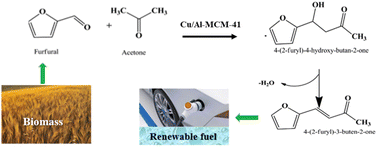
The conversion of biomass into fuels or fuel intermediates is a useful technique to reduce the dependency on fossil fuels. In this study, a liquid fuel intermediate like 4-(2-furyl)-3-buten-2-one (FAc) was produced via an aldol condensation of xylose-derived furfural and acetone over Cu/Al-MCM-41, Cu/Al-TUD-1, and Cu/FDU-12 mesoporous catalysts. The catalysts were characterized by N2-adsorption, NH3-TPD, and pyridine-FTIR for textural properties and acidity, respectively. Among these catalysts, after a reaction time of 24 h at 175 °C and an acetone to furfural mole ratio of 40, the maximum conversion of furfural with the highest selectivity to FAc was obtained with the Cu/Al-MCM-41 catalyst. The process parameters such as the operating time (8 to 45 h), temperature (150 to 200 °C), and acetone to furfural mole ratio (10 to 55) were optimized by Response Surface Methodology (RSM). This model predicted a conversion and product selectivity to FAc of 96 and 90%, respectively, close to the experimentally observed values of 98 and 92%. The synergistic catalytic effect of Lewis and Brønsted acid sites of Cu/Al-MCM-41 on the conversion of furfural and selectivity to FAc was apparent. The catalyst was found to be reusable three times without needing regeneration. After regeneration, the conversion and product selectivity over the Cu/Al-MCM-41 catalyst dropped only by 9 and 3% respectively. The reaction followed a pseudo-first-order kinetic model with an apparent activation energy of 15.5 kJ mol−1. ANOVA analysis was carried out to determine process parameters' interactive effects.


 Please wait while we load your content...
Please wait while we load your content...