Does the presence of lignin affect the pyrolytic decomposition of cellulose? A condensed phase computational investigation†
Abstract
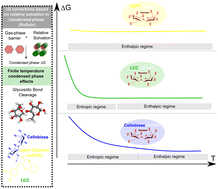
The multiscale nature of biomass and the interplay between its components make it difficult to investigate its pyrolysis chemistry. As the first step in this direction, a first-principles based molecular investigation is performed to elucidate the effect of lignin on the condensed phase activation of cellulose at different temperatures. Key mechanisms for cellulose activation are simulated and reaction energetics in the condensed phase are computed. Lignin presence in the pyrolytic melt is modelled using two condensed phase models – (1) a melt-phase with LCC linkages and (2) a lignin-rich melt-phase with no LCC bonding. Compared to gas phase calculations, free energy barriers for cellobiose activation via transglycosylation in the LCC melt dropped by 100+ kJ mol−1 and activation in the LCC melt phase at temperatures lower than 800 K was kinetically more facile than in the pure cellobiose condensed phase. However, the lignin-rich melt, without LCC, had little impact on cellulose activation, with free energy barriers comparable to those in the gas phase. The breakdown of hydrogen bonds and the change in hydroxymethyl group orientation in cellobiose (due to change in temperature and the condensed phase composition) can be directly mapped to the reaction energetics with the exception of the ring contraction mechanism in the melt-phase with LCC linkages. Thus, the presence of lignin has contrasting effects on cellulose activation–reduction in the activation free energy barrier, in comparison to the pure cellobiose condensed phase, in the covalently bonded LCC melt-phase and relatively higher free energy barriers in the lignin-rich melt-phase, without LCC linkages.


 Please wait while we load your content...
Please wait while we load your content...