PDI–trityl dyads as photogenerated molecular spin qubit candidates†
Abstract
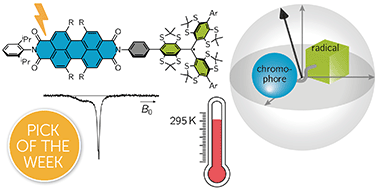
Owing to their potential applications in the field of quantum information science, photogenerated organic triplet–radical conjugates have attracted an increasing amount of attention recently. Typically, these compounds are composed of a chromophore appended to a stable radical. After initialisation of the system by photoexcitation, a highly spin-polarised quartet state may be generated, which serves as a molecular spin qubit candidate. Here, we investigate three perylene diimide (PDI)-based chromophore–radical systems with different phenylene linkers and radical counterparts by both optical spectroscopy and transient electron paramagnetic resonance (EPR) techniques. Femtosecond transient absorption measurements demonstrate chromophore triplet state formation on a picosecond time scale for PDI–trityl dyads, while excited state deactivation is found to be slowed down considerably in a PDI–nitroxide analogue. The subsequent investigation of the coherent spin properties by transient EPR confirms quartet state formation by triplet–doublet spin mixing for all investigated dyads and the suitability of the two studied PDI–trityl dyads as spin qubit candidates. In particular, we show that using tetrathiaryl trityl as the radical counterpart, an intense spin polarisation is observed even at room temperature and quartet state coherence times of 3.0 μs can be achieved at 80 K, which represents a considerable improvement compared to previously studied systems.
- This article is part of the themed collection: 2023 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...