Semi-enzymatic acceleration of oxidative protein folding by N-methylated heteroaromatic thiols†
Abstract
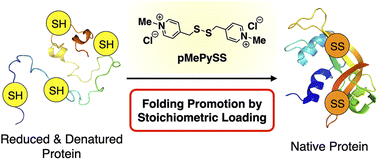
We report the first example of a synthetic thiol-based compound that promotes oxidative protein folding upon 1-equivalent loading to the disulfide bonds in the client protein to afford the native form in over 70% yield. N-Methylation is a central post-translational processing of proteins in vivo for regulating functions including chaperone activities. Despite the universally observed biochemical reactions in nature, N-methylation has hardly been utilized in the design, functionalization, and switching of synthetic bioregulatory agents, particularly folding promotors. As a biomimetic approach, we developed pyridinylmethanethiols to investigate the effects of N-methylation on the promotion of oxidative protein folding. For a comprehensive study on the geometrical effects, constitutional isomers of pyridinylmethanethiols with ortho-, meta-, and para-substitutions have been synthesized. Among the constitutional isomers, para-substituted pyridinylmethanethiol showed the fastest disulfide-bond formation of the client proteins to afford the native forms most efficiently. N-Methylation drastically increased the acidity and enhanced the oxidizability of the thiol groups in the pyridinylmethanethiols to enhance the folding promotion efficiencies. Among the isomers, para-substituted N-methylated pyridinylmethanethiol accelerated the oxidative protein folding reactions with the highest efficiency, allowing for protein folding promotion by 1-equivalent loading as a semi-enzymatic activity. This study will offer a novel bioinspired molecular design of synthetic biofunctional agents that are semi-enzymatically effective for the promotion of oxidative protein folding including biopharmaceuticals such as insulin in vitro by minimum loading.
- This article is part of the themed collection: 2023 Chemical Science Covers


 Please wait while we load your content...
Please wait while we load your content...