Combining native mass spectrometry and lipidomics to uncover specific membrane protein–lipid interactions from natural lipid sources†
Abstract
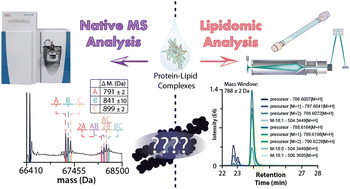
While it is known that lipids play an essential role in regulating membrane protein structure and function, it remains challenging to identify specific protein–lipid interactions. Here, we present an innovative approach that combines native mass spectrometry (MS) and lipidomics to identify lipids retained by membrane proteins from natural lipid extracts. Our results reveal that the bacterial ammonia channel (AmtB) enriches specific cardiolipin (CDL) and phosphatidylethanolamine (PE) from natural headgroup extracts. When the two extracts are mixed, AmtB retains more species, wherein selectivity is tuned to bias headgroup selection. Using a series of natural headgroup extracts, we show TRAAK, a two-pore domain K+ channel (K2P), retains specific acyl chains that is independent of the headgroup. A brain polar lipid extract was then combined with the K2Ps, TRAAK and TREK2, to understand lipid specificity. More than a hundred lipids demonstrated affinity for each protein, and both channels were found to retain specific fatty acids and lysophospholipids known to stimulate channel activity, even after several column washes. Natural lipid extracts provide the unique opportunity to not only present natural lipid diversity to purified membrane proteins but also identify lipids that may be important for membrane protein structure and function.
- This article is part of the themed collection: Most popular 2023 analytical chemistry articles


 Please wait while we load your content...
Please wait while we load your content...