Organocatalyzed enantio- and diastereoselective isomerization of prochiral 1,3-cyclohexanediones into nonalactones bearing distant stereocenters†
Abstract
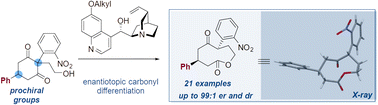
The lactonization of 2-(2-nitrophenyl)-1,3-cyclohexanediones containing an alcohol side chain and up to three distant prochiral elements is reported by isomerization under the mediation of simple organocatalysts such as quinidine. Through a process of ring expansion, strained nonalactones and decalactone are produced with up to three stereocenters in high er and dr (up to 99 : 1). Distant groups, including alkyl, aryl, carboxylate and carboxamide moieties, were examined.


 Please wait while we load your content...
Please wait while we load your content...