Revisiting the Mukaiyama-type epoxidation for the conversion of styrene into styrene carbonate in the presence of O2 and CO2†
Abstract
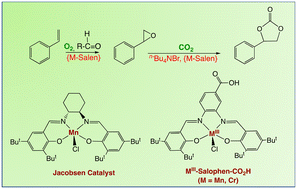
Since its first report in the early 1990s, Mukaiyama-type aerobic epoxidation involving the presence of an aldehyde as an electron donor is considered as an interesting oxidation reaction. Among the various metal catalysts efficient for this process, salen-type complexes have many advantages: i) cheap Schiff-base ligands can be easily synthesized and functionalized, ii) their metal complexes are described with most transition metals from the periodic table and iii) these compounds are also efficient for the catalytic formation of cyclic carbonates from epoxides and CO2, which makes them ideal candidates for the oxidative carboxylation of alkenes. The present work aimed at revisiting the aerobic oxidation of styrene using Cr(III) and Mn(III) salen complexes in the presence of isobutyraldehyde with the idea to further use these complexes (without any work-up), as co-catalysts for the formation of styrene carbonate under CO2 pressure. However, contrary to what is often described in the literature, the present study showed that the introduction of a metal catalyst is not necessary for the epoxidation of styrene at 80 °C in benzonitrile. In our hands, yields of ca. 15% of styrene carbonate were obtained from styrene using O2 (3.5 bar) and CO2 (11 bar) as reagents in the presence of isobutyraldehyde (2.5 eq), tetrabutylammonium bromide (ca. 2 mol%) and the Cr(III) salen complex (ca. 1 mol%). Apparently, the oxidation of Cr(III) to Cr(VI) species in the presence of O2 and isobutyraldehyde is detrimental to the kinetics of the tetrabutylammonium bromide-catalyzed cycloaddition of CO2 to styrene oxide.


 Please wait while we load your content...
Please wait while we load your content...