Mechanism and origins of ligand-controlled regioselectivity of copper-catalyzed borocarbonylation of imines with B2pin2 and alkyl iodides: a computational study†
Abstract
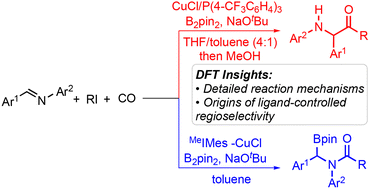
Density functional theory calculations have been performed to investigate the copper-catalyzed borocarbonylation of imines with B2pin2 and alkyl iodides. The computations reveal that the reaction is initiated by the selective migratory insertion of the C–N double bond into the Cu–B bond to forge the key α-boryl amido-copper complex, which could undergo the 1,2-rearrangment via a stepwise dearomatization–rearomatization pathway to give α-amino alkyl-copper species. From these intermediates, the following C–I bond cleavage of alkyl iodide through halogen-atom abstraction, radical association, and reductive elimination would lead to α-boryl amide and α-amino ketone products. The regioselectivity of the overall reaction is determined by the competition between the 1,2-rearrangement and C–I bond cleavage from the α-boryl amido-copper complex. The experimentally observed ligand-controlled regioselectivity is reproduced very well by the computations, which is rationalized on the basis of Cu⋯O interaction and Cu–N bond strength.


 Please wait while we load your content...
Please wait while we load your content...