Glycosyl benzoates as novel substrates for glycosynthases†
Abstract
The development of a procedure for the one-pot synthesis of glycosyl benzoates directly from unprotected sugars in aqueous media using 2-chloro-1,3-dimethylimidazolium chloride (DMC), thiobenzoic acid, and triethylamine is reported. These glycosyl donors are excellent substrates for wild-type and mutant glycosidases. β-Glucosyl benzoate was hydrolysed by the GH1 β-glucosidase derived from Halothermothrix orenii (HorGH1). Subsequent use of this substrate in thioligase-mediated glycosylation of p-nitrothiophenol demonstrated their superiority as donors compared to their p-nitrophenol counterparts with excellent conversions. Using a series of arene nucleophiles, we also demonstrate good to excellent conversions (up to 94%) of β-glucosyl benzoate to the corresponding p-nitrophenyl- and thioglycosides.
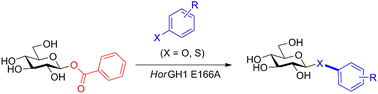


 Please wait while we load your content...
Please wait while we load your content...