One-pot domino synthesis of five- and six-membered fused dihydropyridines promoted by PPh3–NBS in aqueous medium†
Abstract
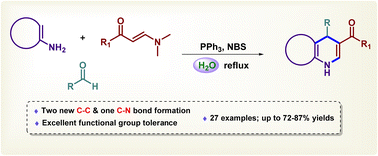
A facile one-pot synthesis of five- and six-membered fused dihydropyridines such as chromenodihydropyridines, pyrazolodihydropyridines and isoxazolopyridines was accomplished for the first time by employing PPh3–NBS via a formal [3 + 2 + 1] cycloaddition of 1,3-bisnucleophiles (i.e., 2-aminochromone, 4-aminochromone, 5-aminopyrazole and 5-aminoisoxazole), β-enaminones and aldehydes in aqueous medium. The present approach involves a Michael type addition followed by intramolecular cyclization leading to the formation of two new C–C bonds and one C–N bond. High compatibility and excellent yields are the advantages of this protocol.


 Please wait while we load your content...
Please wait while we load your content...