Anode-free Na metal batteries developed by nearly fully reversible Na plating on the Zn surface†
Abstract
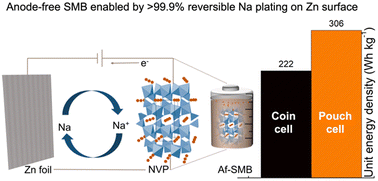
The anode-free battery architecture has recently emerged as a promising platform for lithium and sodium metal batteries as it not only offers the highest possible energy density, but also eliminates the need for handling hazardous metal electrodes during cell manufacturing. However, such batteries usually suffer from much faster capacity decay and are much more sensitive to even trace levels of irreversible side reactions on the anode, especially for the more reactive Na metal. This work systematically investigates electrochemical interfaces for Na plating and stripping and describes the use of the Zn surface to develop nearly fully reversible Na anodes with 1.0 M NaPF6 in a diglyme-based electrolyte. The high performance includes consistently higher than 99.9% faradaic efficiencies for a wide range of cycling currents between 0.5 and 10 mA cm−2, much more stable interfacial resistance and nearly no formation of mossy Na after 500 cycles compared with conventional Al and Cu surfaces. This improved reversibility was further confirmed under lean electrolyte conditions with a wide range of electrolyte concentrations and cycling temperatures and can be attributed to the strong interfacial binding and intrinsic sodiophilic properties of the Zn surface with Na, which not only ensured uniform Na plating but also eliminated most side reactions that would otherwise cause electrolyte depletion. As a result, full cells assembled with Na-free Zn foil and a high capacity Na3V2(PO4)3 cathode delivered ∼90% capacity retention for 100 cycles, higher than the 73% retention of Cu foils and much higher than the 39% retention of Al foils. This work provides new approaches to enable stable cycling of anode-free batteries and contribute to their applications in practical devices.
- This article is part of the themed collections: Nanoscale Most Popular 2023 Articles and Nanoscale 2023 Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...