Dissolution profile prediction of sinomenine hydrochloride sustained release tablets, part II: a multisource data fusion approach
Abstract
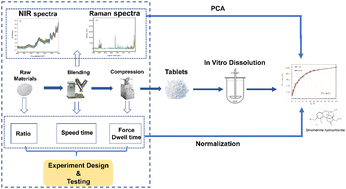
The aim of this study was to verity the feasibility of in vitro dissolution behavior prediction using a multi-source data fusion strategy, taking the prepared sinomenine hydrochloride (SH) sustained release tablets as examples. In this work, 180 batches of mixed powders of SH sustained release tablets were scanned by near-infrared (NIR) spectroscopy and Raman spectroscopy, respectively. The tablets were then prepared using an experimental design, which consisted of a 3-level full factorial design and a D-optimal design. After obtaining the in vitro dissolution profiles of SH sustained release tablets, prediction models were developed using the partial least squares (PLS) regression algorithm based on NIR spectra or Raman spectra individually. Subsequently, data fusion models were built based on optimal principal components (PCs) combined with formulation variables and process parameters using the PLS regression and back-propagation artificial neural network (BP-ANN) algorithms, respectively. The results of the PLS regression model established by the data fusion strategy indicated better performance than the prediction models built using spectra individually. The value of variable importance for projection (VIP) indicated that the content of hydroxypropyl methylcellulose (HPMC) was the main factor influencing the dissolution behavior of SH sustained release tablets. The results of the difference factor (f1) and similarity factor (f2), as well as the paired t-test, for the parameters of the fitted equations indicated that there was no difference between the measured and predicted values of dissolution profiles of the validation set samples. In summary, the data fusion strategy established in this work can provide a solution for the quality evaluation of sustained release tablets in a rapid way.


 Please wait while we load your content...
Please wait while we load your content...