Cobalt-doped copper vanadate: a dual active electrocatalyst propelling efficient H2 evolution and glycerol oxidation in alkaline water†
Abstract
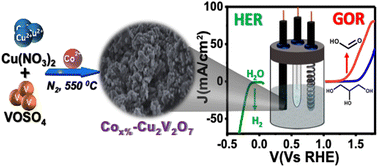
Strategically doped metal oxide nanomaterials signify a rapidly growing genre of functional materials with a wide range of practical applications. Copper vanadate (CuV) represents one such highly active system, which has been rarely explored following its doping with an abundant first-row transition metal. Here, we have developed a series of CuV samples with varying cobalt(II) doping concentrations deploying a relatively simple solid state synthetic procedure. Among the samples, the 10% Co(II)-doped CuV (Co10%–CuV) exhibited excellent reactivity for both the H2 evolution reaction (HER) and glycerol oxidation reaction (GOR) in an alkaline aqueous medium (pH 14.0) during cathodic and anodic scans, respectively. During this dual-active catalysis, surface-immobilized Co10%–CuV operates at exceptionally low overpotentials of 176 mV and 160 mV for the HER and GOR, respectively, while achieving 10 mA cm2 current density. The detailed spectroscopic analysis revealed the formation of formate as the major product during the GOR with a faradaic efficiency of >90%. Therefore, this Co10%–CuV can be included on either side of a two-electrode electrolyzer assembly to trigger a complete biomass-driven H2 production, establishing an ideal carbon-neutral energy harvest process.
- This article is part of the themed collections: Celebrating nanoscience in India and Popular Advances


 Please wait while we load your content...
Please wait while we load your content...