Environmental evaluation of carbon dot and fullerene formation in atmospheric particulate matter (PM10): observations from their free radical scavenging and electrochemical studies†
Abstract
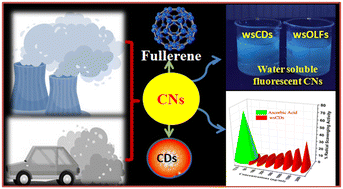
Human exposure to atmospheric particulate matter (PM) is of great interest owing to its carbonaceous nature with potential health impact. Carbonaceous particulate matter (PM) is mainly composed of black carbon and organic carbon. The abundance of carbon in atmospheric PM can also lead to the formation of carbon nanomaterials under different environmental conditions. In this innovative work, we evaluated the formation of water-soluble carbon dots (wsCDs) and onion-like fullerenes (wsOLFs) from atmospheric particulate matter (PM) in an oxidizing environment. The synthesized wsCDs were quasi-spherical in shape with un-agglomerated or uniformly distributed particles with an average size of 4–6 nm. The wsOLFs were spherical and/or quasi-spherical in shape, with corresponding particle size distribution of an average diameter ranging from 8–12 nm. High resolution transmission electron microscopy images also showed concentric spherical shells like onions and fringes of graphitic planes with interplanar distances of 0.232–0.297 nm, which are smaller than that of graphite (0.336 nm). The PM-derived carbon dots and fullerenes were observed to be effective antioxidants from their free radical scavenging studies. Further, promising electrochemical properties are also detected with these materials indicating their potential prospects for development of environmental nanotechnology.


 Please wait while we load your content...
Please wait while we load your content...