pH affects the aqueous-phase nitrate-mediated photooxidation of phenolic compounds: implications for brown carbon formation and evolution†
Abstract
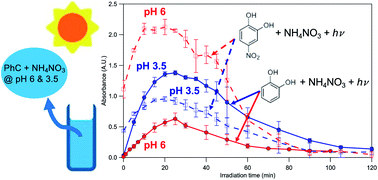
Brown carbon (BrC) is known to have important impacts on atmospheric chemistry and climate. Phenolic compounds are a prominent class of BrC precursors that are emitted in large quantities from biomass burning and fossil fuel combustion. Inorganic nitrate is a ubiquitous component of atmospheric aqueous phases such as cloudwater, fog, and aqueous aerosols. The photolysis of inorganic nitrate can lead to BrC formation via the photonitration of phenolic compounds in the aqueous phase. However, the acidity of the atmospheric aqueous phase adds complexity to these photonitration processes and needs to be considered when investigating BrC formation from the nitrate-mediated photooxidation of phenolic compounds. In this study, we investigated the influence of pH on the formation and evolution of BrC from the aqueous-phase photooxidation of guaiacol, catechol, 5-nitroguaiacol, and 4-nitrocatechol initiated by inorganic nitrate photolysis. The reaction rates, BrC composition and quantities were found to depend on the aqueous phase pH. Guaiacol, catechol, and 5-nitroguaiacol reacted substantially faster at lower pH. In contrast, 4-nitrocatechol reacted at slower rates at lower pH. For all four phenolic compounds, the initial stages of photooxidation resulted in an increase in light absorption (i.e., photo-enhancement) in the near-UV and visible range due to the formation of light absorbing products formed via the addition of nitro and/or hydroxyl groups to the phenolic compound. Greater photo-enhancement was observed at lower pH during the nitrate-mediated photooxidation of guaiacol and catechol. In contrast, greater photo-enhancement was observed at higher pH during the nitrate-mediated photooxidation of 5-nitroguaiacol and 4-nitrocatechol. This indicated that the effect that the aqueous phase pH has on the composition and yields of BrC formed is not universal, and will depend on the initial phenolic compound. These results provide new insights into how the atmospheric aqueous phase acidity influences the reactivities of different phenolic compounds and BrC formation/evolution during photooxidation initiated by inorganic nitrate photolysis, which will have significant implications for how the atmospheric fates of phenolic compounds and BrC formation/evolution are modeled for areas with high levels of inorganic nitrate.
- This article is part of the themed collections: Atmospheric chemistry and Chemistry of Atmospheric Pollutants


 Please wait while we load your content...
Please wait while we load your content...