Human skin oil: a major ozone reactant indoors†
Abstract
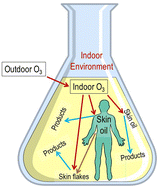
Human skin oil is rich in ozone-reactive compounds, including squalene and unsaturated acyl groups of free fatty acids, glycerols, and wax esters. Squalene and unsaturated acyl groups are each responsible for about half of the double bonds in skin oil. When there are no indoor sources, ozone concentrations are smaller indoors than outdoors, chiefly because ozone reacts with indoor surfaces. Ozone reacts rapidly with skin oils on occupants' exposed skin, hair, and clothing. Also, skin oil and its unsaturated oxidation products are transferred to indoor surfaces. A recent study of an occupied residence inferred that the average surface density of skin oil double bonds on inanimate indoor surfaces was approximately 5 μmol m−2. Estimates suggest that about 15% of outdoor ozone transported into residences is removed by skin oil or its byproducts. This percentage increases with occupant density indoors. In classrooms, the proportion of ozone removal attributable to skin oil may be in the range 35–55%. Further measurements of skin oil on off-body surfaces in a variety of indoor environments are needed to improve such estimates. In occupied indoor environments, the amount of ozone and ozone-derived products that occupants inhale is materially affected by the extent to which ozone reacts with skin-oil constituents. Toxicities of the products of ozone reactions with skin oil warrant further attention. The relative contribution of other fugitive sources (e.g., cooking oils, paints, and pesticides), as well as constituents inherent to building materials and furnishings, to ozone-reactive compounds on indoor surfaces remains unknown and also merits attention.
- This article is part of the themed collections: The role of tropospheric ozone in atmospheric processes, health and climate - Topic Highlight, International Day for the Preservation of the Ozone Layer, Outstanding Papers 2023 – Environmental Science: Atmospheres, Outstanding Papers of 2023 from RSC’s Environmental Science journals and Indoor Air Quality


 Please wait while we load your content...
Please wait while we load your content...