NacNac-zinc-pyridonate mediated ε-caprolactone ROP†
Abstract
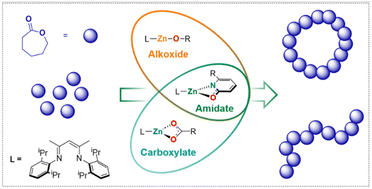
Herein we report the synthesis, isolation and polymerisation activity of two new zinc compounds based on a 2,6-diisopropylphenyl (Dipp) β-diiminate (NacNac) ligand framework with zinc also ligated by an amidate (2-pyridonate or 6-methyl-2-pyridonate) unit. The compounds crystallised as either monomeric (6-Me-2-pyridonate derivative) or dimeric (2-pyridonate) species, although both were found to be monomeric in solution via1H DOSY NMR spectroscopy, which was supported by DFT calculations. These observations suggest that both complexes initiate ring-opening polymerisation (ROP) through a single-site monometallic mechanism. High molecular weight poly ε-caprolactone (PCL) was achieved via exogenous initiator-free ROP conditions with both catalysts. An increase in the 2-pyridonate initiator steric bulk (6-Me- vs. 6-H-) resulted in an improved catalytic activity, facilitating complete monomer conversion within 1 h at 60 °C. Pyridonate end-groups were observed by MALDI-ToF mass spectrometry, contrasting with previous observations for DippNacNac-Zn acetate complexes (where no acetate end groups are observed), instead this more closely resembles the reactivity of DippNacNac-Zn alkoxide complexes in ROP (where RO end groups are observed). Additional major signals in the MALDI-ToF spectra were consistent with cyclic PCL species, which are attributed to back-biting ring-closing termination steps occuring in a process facilitated by the pyridonate unit being an effective leaving group. To the best of our knowledge, these complexes represent the first examples of pyridonate, and indeed amidate, initated ROP.


 Please wait while we load your content...
Please wait while we load your content...