Carbon dioxide sequestration in natural gas hydrates – effect of flue and noble gases†
Abstract
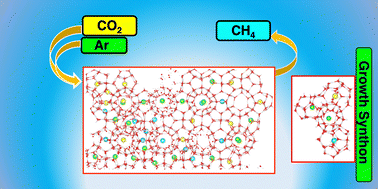
Clean energy is one of the immediate requirements all over the world to tackle the global energy demands. Natural gas hydrates (NGHs) are one of the proposed alternatives that could be used to extract methane as clean energy and simultaneously sequestrate carbon dioxide. However, the formation of CH4–CO2 mixed hydrates and the first hydrate layer besides the interface reduces the rate of CO2 sequestration and methane extraction in NGHs, and thus, multistep extraction of methane is one of the proposed solutions. We report the atomic level factors that could enhance CO2 sequestration in the newly formed first hydrate layer besides the interface in the presence of flue and noble gases using DFT calculations and molecular dynamics simulations at 250 K and 0.15 kbar. The simulations show the formation of stable dual cages (large-large or small-large) that lead to the formation of a four-caged, Y-shaped cluster (growth synthon) which leads to the formation of a hydrate unit cell in heterogeneous medium. Among the flue and noble gases, only argon forms energetically favorable dual cages with itself and CO2 due to which enhanced CO2 sequestration is observed at different concentrations of Ar and CO2 where the CO2 : Ar (2.5 : 1.5) system shows the best CO2 sequestration in the first layer besides the interface. The results also provide understanding into the previously reported concentration dependent CO2 selectivity in sI hydrates in the presence of third gases (N2 and H2S).


 Please wait while we load your content...
Please wait while we load your content...