How water desorbs from calcite†
Abstract
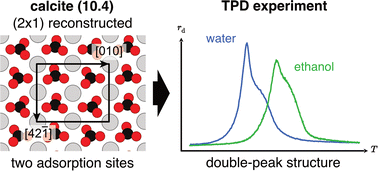
Calcite is the most abundant carbonate mineral in Earth's crust. Upon cleavage, the (10.4) plane with a rectangular unit cell is exposed. Interestingly, several experiments suggest a (2 × 1) surface reconstruction. However, clear experimental evidence and a theoretical confirmation were long missing. Recently, convincing indication for a (2 × 1) reconstruction has been given by atomic force microscopies taken at 5 K. Here, we show temperature-programmed desorption (TPD) experiments of water and ethanol desorbing from calcite (10.4) around room temperature. The TPD curves fit excellently to a kinetic model considering two different adsorption sites, as expected in case of a (2 × 1) reconstruction. This finding applies to the desorption of water and ethanol, illustrating that the effect is characteristic for the calcite cleavage plane. Our results thus show that the (2 × 1) reconstruction not only exists at room temperature but has significant impact on the interfacial properties of calcite.


 Please wait while we load your content...
Please wait while we load your content...