Mechanism of rate controllability of water-soluble bifunctional cyclooctadiynes through cation-anion interactions†
Abstract
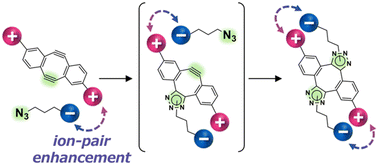
Click reactions are used for chemoselective functionalization in many research fields. Despite the utility of small, bioinert azide groups as a counterpart, applications of strain-promoted alkyne-azide cycloaddition (SPAAC) reactions for this purpose are still limited by slow reaction kinetics. Here, we report ion-pair-guided reaction rate enhancement by the use of water-soluble cyclooctadiynes (WS-CODYs) composed of bifunctional strained alkynes and polar side chains. Arrhenius plot analysis revealed that the rate enhancement by WS-CODYs is due to a kinetic salt effect between the polar substituents and the target azide. We demonstrate the utility of these compounds for rapid protein labelling and isoelectric point-dependent labelling.


 Please wait while we load your content...
Please wait while we load your content...