Efficient generation of highly crystalline carbon quantum dots via electrooxidation of ethanol for rapid photodegradation of organic dyes†
Abstract
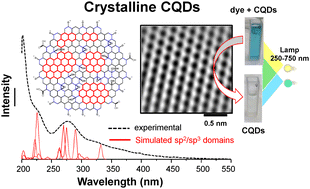
Achieving versatile routes to generate crystalline carbon-based nanostructures has become a fervent pursuit in photocatalysis-related fields. We demonstrate that the direct electrooxidation of ethanol, performed on Ni foam, yields ultra-small and highly crystalline graphene-like structures named carbon quantum dots (CQDs). We perform simulations of various sp2 and sp3 domains in order to understand the optical properties of CQDs by accounting their contribution as absorbance/luminescent centers in the overall optical response. Experiments and simulations reveal that absorbance bands for as-synthesized CQDs are dominated by small sp2 domains comprised of ≤7 aromatic-rings. After 48 h synthesis, the dispersion transition from yellow to red, exhibiting new and red shifted absorbance bands. Furthermore, fluorescence emission is governed by medium-sized sp2 domains (with aromatic ring counts ≤12) and oxygen-containing groups. These oxygen-rich groups within the CQDs, confirmed by FT-IR and XPS, are responsible for the fast photodegradation of organic dyes, with ∼90% of methylene blue (MB) being degraded within the first 5 min of light exposure. Our work provides crucial insights about the electrochemical synthesis and overall optical properties of carbon nanostructures, while being effective and reliable toward the degradation of contaminants in water.
- This article is part of the themed collection: #MyFirstJMCC


 Please wait while we load your content...
Please wait while we load your content...