The unexpected photoelectrochemical activity of MAX phases: the role of oxide impurities†
Abstract
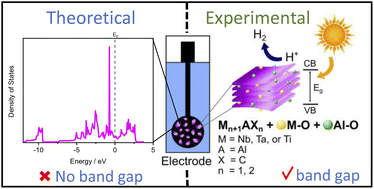
MAX phases are layered ternary compounds that are mainly studied for their physical properties and their use in the synthesis of MXenes. Their application in energy generation has been investigated and recently, the unexpected photoactivity of MAX phases under the influence of a visible light source has been reported. To investigate the origin of this photoactivity, theoretical calculations and experimental characterisation of the structural and optical properties of three MAX phases, Nb2AlC, Ta2AlC and Ti3AlC2, were performed. Although the theoretical calculations confirmed that the phases presented no band gap in the vicinity of the Fermi level, the experimental evaluation showed two main absorptions for Nb2AlC and Ta2AlC (2.2 eV, 3.1 eV, and 2.2 eV, 3.3 eV, respectively) and one for Ti3AlC2 (2.4 eV). To confirm the observations from the optical characterisation, the phases were applied as photoelectrocatalysts for hydrogen generation under the influence of light of different wavelengths. Nb2AlC and Ta2AlC performed better when exposed to UV light, while Ti3AlC2 showed the lowest overpotential under the influence of visible light, in accordance with the experimentally estimated band gaps. The materials were extensively characterised and the photoactivity of MAX phases was attributed to the presence of photoactive oxide impurities on the surface of the material, which are naturally formed from contact with air and solvents. In this work, we show how these impurities can lead to better performances thanks to their intrinsic photoactivity, indicating the prospects for the use of MAX phases in other photoelectrochemical processes.


 Please wait while we load your content...
Please wait while we load your content...