Tailoring lixiviant properties to optimise selectivity in E-waste recycling†
Abstract
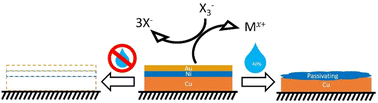
The scale of E-waste production makes the selective recovery of technology metals an important research topic. It has previously been shown that deep eutectic solvents, DESs, can be used to rapidly digest gold and in the current study the effect of varying water and ethylene glycol content on the ability to selectively recover metals in DESs was investigated. It was found that increased water content resulted in an increase in metal etching rates for copper due to the decreasing viscosity of the solution, but etching rates of nickel, silver and gold were decreased due to the competition between chloride and oxide/hydroxide chemistry causing passivating films to form. Iodine was used as a catalyst and speciation was investigated in different solvent compositions. The ratio of the two trihalide species was mostly unaffected by solvent composition, but the presence of molecular iodine was detected with 20 wt% water due to decreased iodine solubility resulting from lower chloride concentration. It was determined that solvent physical properties were most important for metal etching rates at low water content, but at high water content the effect of oxide/hydroxide chemistry was more significant, resulting in the possibility of selective metal etching of copper from PCBs.


 Please wait while we load your content...
Please wait while we load your content...