A π-conjugated covalent organic framework enables interlocked nickel/photoredox catalysis for light-harvesting cross-coupling reactions†
Abstract
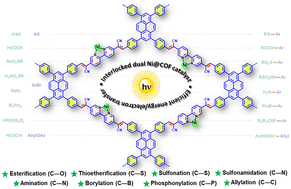
Covalent organic frameworks (COFs) are an outstanding platform for heterogeneous photocatalysis. Herein, we synthesized a pyrene-based two-dimensional C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C linked π-conjugated COF via Knoevenagel condensation and anchored Ni(II)-centers through bipyridine moieties. Instead of traditional dual metallaphotoredox catalysis, the mono-metal decorated Ni@Bpy-sp2c-COF interlocked the catalysis mediated by light and the transition metal. Under light irradiation, enhanced energy and electron transfer in the COF backbone, as delineated by the photoluminescence, electrochemical, and control experiments, expedited the excitation of Ni centers to efficiently catalyze diverse photocatalytic C–X (X = B, C, N, O, P, S) cross-coupling reactions with efficiencies orders of magnitude higher than the homogeneous controls. The COF catalyst tolerated a diverse range of coupling partners with various steric and electronic properties, delivering the products with up to 99% yields. Some reactions were performed on a gram scale and were applied to diversify pharmaceuticals and complex molecules to demonstrate the synthetic utility.
C linked π-conjugated COF via Knoevenagel condensation and anchored Ni(II)-centers through bipyridine moieties. Instead of traditional dual metallaphotoredox catalysis, the mono-metal decorated Ni@Bpy-sp2c-COF interlocked the catalysis mediated by light and the transition metal. Under light irradiation, enhanced energy and electron transfer in the COF backbone, as delineated by the photoluminescence, electrochemical, and control experiments, expedited the excitation of Ni centers to efficiently catalyze diverse photocatalytic C–X (X = B, C, N, O, P, S) cross-coupling reactions with efficiencies orders of magnitude higher than the homogeneous controls. The COF catalyst tolerated a diverse range of coupling partners with various steric and electronic properties, delivering the products with up to 99% yields. Some reactions were performed on a gram scale and were applied to diversify pharmaceuticals and complex molecules to demonstrate the synthetic utility.
- This article is part of the themed collection: Most popular 2023 catalysis articles


 Please wait while we load your content...
Please wait while we load your content...