Anti-myeloma pro-apoptotic Pt(ii) diiodido complexes†
Abstract
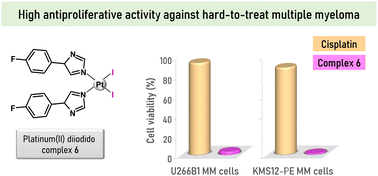
Platinum-based agents unwaveringly hold their prominent position in cancer treatment. Current research emphasizes finding novel complexes for hard-to-treat cancers with minimum side effects, capable of overcoming resistance. This work presents easy-to-prepare diiodidoplatinum(II) complexes cis-[PtI2(Ln)2] (1–7) with imidazole derivatives (Ln), which were considerably effective against multiple myeloma cell lines U266B1 and KMS12-PE. The leading compound 6 (at 3 μM concentration) extraordinarily reduced viability of U266B1 and KMS12-PE myeloma cells to 3.0% and 1.1%, respectively, and exceeded the conventional platinum-based anticancer drug cisplatin (93.1% and 88.3%, respectively) that is used clinically for the combination therapy of multiple myeloma. Complex 6 was significantly more effective in inducing apoptosis in KMS12-PE cells without interleukin-6 (IL-6) expression than in U266B1 cells with IL-6 expression. Complex 6 also induced apoptosis in co-culture of KMS12-PE with non-cancerous stromal fibroblasts (HS-5), and displayed markedly lower activity in the HS-5 stromal fibroblast cells than in myeloma cells, pointing out its pharmocologically prospective selectivity towards the cancer cells over the normal ones. No caspase 3/7 activity was detected in apoptotic KMS12-PE cells treated by complex 6 indicating a different mechanism of apoptosis action from cisplatin. This work demonstrates that simple non-classical platinum(II) complexes represent a new perspective for a monotherapy of hard-to-treat multiple myeloma.


 Please wait while we load your content...
Please wait while we load your content...