Towards potential antifungal agents: synthesis, supramolecular self-assembly and in vitro activity of azole mono-, sesqui- and diterpenoids†
Abstract
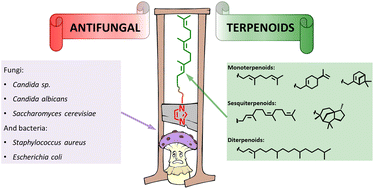
Terpenes and their derivatives are natural antifungal and antimicrobial agents. In this paper, potential antifungal agents were developed on the basis of farnesol, geraniol, myrtenol, perillyl alcohol, cedrol and phytol. The synthesized compounds exist in aqueous solutions as stable associates (D = 142–216 nm, PDI 0.04–0.16, Z = +0.9–+46 mV). Formation of stable associates of the compounds in solution promotes compaction and dosed release of the drug. The membranotropic activity of the compounds was also investigated to open up their possible application in the treatment of skin diseases. The relationship between membranotropism and lipophilicity coefficient (log P) has been established. The antifungal and antimicrobial activities of the obtained compounds were studied in vitro on the clinical isolate of Candida sp., Candida albicans, yeast Saccharomyces cerevisiae, Gram-negative (Escherichia coli) and Gram-positive (Staphylococcus aureus, Bifidobacterium bifidum) bacteria. Candida sp. (0.0781 mg mL−1) and Saccharomyces cerevisiae (0.0049 mg mL−1) showed the highest sensitivity to the agents.


 Please wait while we load your content...
Please wait while we load your content...