Unveiling the formation mechanism of the biphenylene network†
Abstract
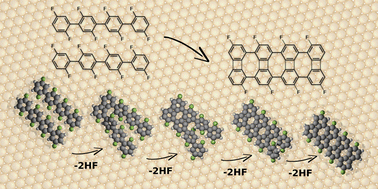
We have computationally studied the formation mechanism of the biphenylene network via the intermolecular HF zipping, as well as identified key intermediates experimentally, on the Au(111) surface. We elucidate that the zipping process consists of a series of defluorinations, dehydrogenations, and C–C coupling reactions. The Au substrate not only serves as the active site for defluorination and dehydrogenation, but also forms C–Au bonds that stabilize the defluorinated and dehydrogenated phenylene radicals, leading to “standing” benzyne groups. Despite that the C–C coupling between the “standing” benzyne groups is identified as the rate-limiting step, the limiting barrier can be reduced by the adjacent chemisorbed benzyne groups. The theoretically proposed mechanism is further supported by scanning tunneling microscopy experiments, in which the key intermediate state containing chemisorbed benzyne groups can be observed. This study provides a comprehensive understanding towards the on-surface intermolecular HF zipping, anticipated to be instructive for its future applications.
- This article is part of the themed collection: Nanoscale and Nanoscale Horizons: Carbon-based nanomaterials


 Please wait while we load your content...
Please wait while we load your content...