Rapid, room-temperature, solvent-free mechanochemical oxidation of elemental gold into organosoluble gold salts†
Abstract
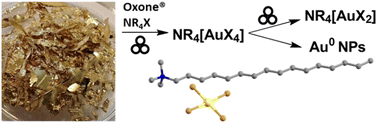
Gold is highly valued for a wide range of commercial and technological applications but is processed exclusively through highly aggressive and toxic solvents and/or reagents, ultimately yielding water-soluble salts that are difficult to separate from inorganic reaction byproducts. As a result, development of safer, cleaner processes that would enable gold processing in non-aqueous, organic solvent is an attractive technological goal. Here, we describe a methodology that simultaneously avoids aggressive reagents and enables gold extraction into a safe organic solvent. The methodology is based on solventless, mechanochemical oxidation of metallic gold with Oxone® in the presence of tetraalkylammonium halide salts, to directly, rapidly (within 30–60 minutes) and at room temperature convert gold metal into solid salts that are immediately soluble in pure organic solvents and aqueous alcoholic media. The organosoluble gold salts are easily separated from sulfate byproducts by direct extraction into the benign solvent ethyl acetate, which is also easily recycled for re-use, providing a strategy for gold activation and dissolution without any additional reagents for purification, such as cation exchange resins, salts, or chelating agents. Besides enabling direct extraction of gold into an organic solvent, the mechanochemically obtained organosoluble gold salts can also be readily used for further syntheses, as shown here by a two-step one-pot route to prepare air- and moisture-resistant Au(I) salts, and an improved synthesis of gold nanoparticles from bulk gold.
- This article is part of the themed collection: 2023 Green Chemistry Hot Articles


 Please wait while we load your content...
Please wait while we load your content...