Local hydrophobicity allows high-performance electrochemical carbon monoxide reduction to C2+ products†
Abstract
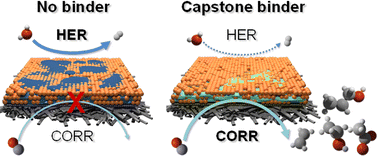
While CO can already be produced at industrially relevant current densities via CO2 electrolysis, the selective formation of C2+ products seems challenging. CO electrolysis, in principle, can overcome this barrier, hence forming valuable chemicals from CO2 in two steps. Here we demonstrate that a mass-produced, commercially available polymeric pore sealer can be used as a catalyst binder, ensuring high rate and selective CO reduction. We achieved above 70% faradaic efficiency for C2+ products formation at j = 500 mA cm−2 current density. As no specific interaction between the polymer and the CO reactant was found, we attribute the stable and selective operation of the electrolyzer cell to the controlled wetting of the catalyst layer due to the homogeneous polymer coating on the catalyst particles’ surface. These results indicate that sophistically designed surface modifiers are not necessarily required for CO electrolysis, but a simpler alternative can in some cases lead to the same reaction rate, selectivity and energy efficiency; hence the capital costs can be significantly decreased.


 Please wait while we load your content...
Please wait while we load your content...