Multiple and nonlocal cation redox in Ca–Ce–Ti–Mn oxide perovskites for solar thermochemical applications†
Abstract
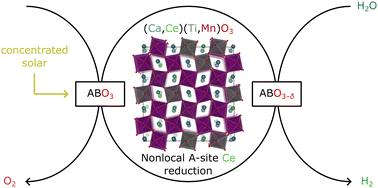
Modeling-driven design of redox-active off-stoichiometric oxides for solar thermochemical H2 production (STCH) seldom has resulted in empirical demonstration of competitive materials. We report the theoretical prediction and experimental evidence that the perovskite Ca2/3Ce1/3Ti1/3Mn2/3O3 is synthesizable with high phase purity, stable, and has desirable redox thermodynamics for STCH, with a predicted average neutral oxygen vacancy (VO) formation energy, Ev = 3.30 eV. Flow reactor experiments suggest potentially comparable or greater H2 production capacity than recent promising Sr–La–Mn–Al and Ba–Ce–Mn metal oxide perovskites. Utilizing quantum-based modeling of a solid solution on both A and B sub-lattices, we predict the impact of nearest-neighbor composition on Ev and determine that A-site Ce4+ reduction dominates the redox-activity of Ca2/3Ce1/3Ti1/3Mn2/3O3. X-ray absorption spectroscopy measurements provide evidence that supports these predictions and reversible Ce4+-to-Ce3+ reduction. Our models predict that Ce4+ reduces even when it is not nearest-neighbor to the VO, suggesting that refinement of Ce stoichiometry has the possibility of further enhancing performance.
- This article is part of the themed collection: Spotlight on Women in Energy


 Please wait while we load your content...
Please wait while we load your content...