Pyridine modifications regulate the electronics and reactivity of Fe–pyridinophane complexes†
Abstract
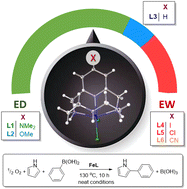
12-Membered pyridinophanes are the focus of many studies as biological mimics, chelators, and catalytic precursors. Therefore, the desire to tune the reactivity of pyridinophanes to better control the applications of derivative metal complexes has inspired many structure–activity relationship studies. However, the separation of structural versus electronic changes imparted by ligand modification has made these structure–activity relationship studies of transition metal catalysts challenging to define. In this work we show that 4-substitution of the pyridine ring in 12-membered tetra-aza pyridinophanes successfully provides a regulatory handle on the electronic properties of the metal center and, therefore, the catalytic C–C coupling activity of the respective iron complexes. The C–C coupling reaction catalyzed by Fe(L1–L6) provides a range of yields (32–58%) that directly correlate with iron redox potentials (ΔE1/2 = 152 mV) and metal binding constants (Δlog β = 3.45), while the geometry of the complexes was virtually indistinguishable. These are the first results to definitively show the redox potential and metal binding as independent properties from the coordination chemistry in one ligand series. Adjustments to these chemical properties were then shown to provide a regulatory handle for the C–C coupling reactivity tuned via pyridine substitution in pyridinophanes.


 Please wait while we load your content...
Please wait while we load your content...