Doubly ionized OCS bond rearrangement upon fragmentation – experiment and theory†
Abstract
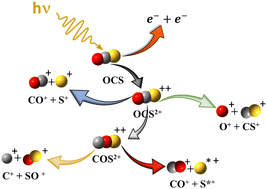
The dissociation of OCS2+ ions formed by photoionization of the neutral molecule at 40.81 eV is examined using threefold and fourfold electron–ion coincidence spectroscopy combined with high level quantum chemical calculations on isomeric structures and their potential energy surfaces. The dominant dissociation channel of [OCS]2+ is charge separation forming CO+ + S+ ion pairs, found here to be formed with low intensity at a lower-energy onset and with a correspondingly smaller kinetic energy release than in the more intense higher energy channel previously reported. We explain the formation of CO+ + S+ ion pairs at low as well as higher ionization energies by the existence of two predissociation channels, one involving a newly identified COS2+ metastable state. We conclude that the dominant CO+ + S+ channel with 5.2 eV kinetic energy release is reached upon OCS2+ → COS2+ isomerization, whereas the smaller kinetic energy release (of ∼4 eV) results from the direct fragmentation of OCS2+ (X3Σ−) ions. Dissociation of the COS2+ isomer also explains the existence of the minor C+ + SO+ ion pair channel. We suggest that isomerization prior to dissociation may be a widespread mechanism in dications and more generally in multiply charged ion dissociations.
- This article is part of the themed collections: Molecular Dynamics in the Gas Phase and 2023 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...