How does thickness affect magnetic coupling in Ti-based MXenes†
Abstract
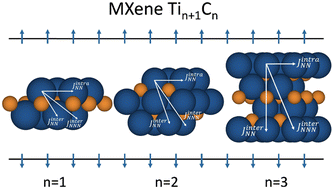
The magnetic nature of Ti2C, Ti3C2, and Ti4C3 MXenes is determined from periodic calculations within density functional theory and using the generalized gradient approximation based PBE functional, the PBE0 and HSE06 hybrids, and the on-site Hubbard corrected PBE+U one, in all cases using a very tight numerical setup. The results show that all functionals consistently predict a magnetic ground state for all MXenes, with spin densities mainly located at the Ti surface atoms. The analysis of solutions corresponding to different spin orderings consistently show that all functionals predict an antiferromagnetic conducting ground state with the two ferromagnetic outer (surface) Ti layers being antiferromagnetically coupled. A physically meaningful spin model is proposed, consistent with the analysis of the chemical bond, with closed shell, diamagnetic, Ti2+ like ions in inner layers and surface paramagnetic Ti+ like centers with one unpaired electron per magnetic center. From a Heisenberg spin model, the relevant isotropic magnetic coupling constants are extracted from an appropriate mapping of total energy differences per formula unit to the expected energy values of the spin Hamiltonian. While the numerical values of the magnetic coupling constants largely depend on the used functional, the nearest neighbor intralayer coupling is found to be always ferromagnetic, and constitutes the dominant interaction, although two other non-negligible interlayer antiferromagnetic terms are involved, implying that the spin description cannot be reduced to NN interaction only. The influence of the MXene thickness is noticeable for the dominant ferromagnetic interaction, increasing its value with the MXene width. However, the interlayer interactions are essentially due to the covalency effects observed in all metallic solutions which, as expected, decay with distance. Within the PBE+U approach, a U value of 5 eV is found to closely simulate the results from hybrid functionals for Ti2C and less accurately for Ti3C2 and Ti4C3.


 Please wait while we load your content...
Please wait while we load your content...