Crystal structures of 5-bromo-1-arylpyrazoles and their halogen bonding features†
Abstract
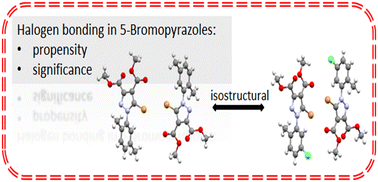
Single crystal X-ray diffraction analysis was employed to investigate the supramolecular properties of five 5-brominated pyrazoles in order to evaluate the role of the Br atom in the formation of halogen bonding duly recognizing that the Br atom is less polarizable than the iodine atom. Hirshfeld analysis was employed to conceive a more descriptive image of the halogen bond propensity of 5-bromopyrazoles. The compounds display Br⋯O contacts or Br⋯Br as type I or type II halogen contacts. The structural analyses revealed the isostructurality of compounds 2 and 3 while compound 5 was found to be isostructural with its iodo-analogue, suggesting the importance of the halogen contacts even if they are considered weak in nature.


 Please wait while we load your content...
Please wait while we load your content...