Cu substitution boosts self-trapped exciton emission in zinc-based metal halides for sky-blue light-emitting diodes†
Abstract
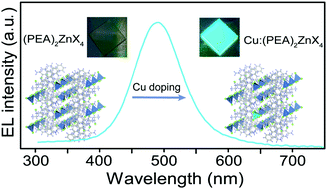
Substantial progress has been made in light-emitting diodes (LEDs) based on red- and green-emitting lead halides. However, reducing the lead toxicity of emissive materials and achieving high-efficiency blue-emitting LED beads on lead-free metal halides remains a great challenge. Herein, lead-free zero-dimensional metal halides Cu:(PEA)2ZnX4 (X = Cl, Br) were developed to solve the above bottlenecks. Cu substitution enhanced the photoluminescence quantum yield (PLQY) of (PEA)2ZnBr4 from 3.4% to 55% and that of (PEA)2ZnCl4 from 2.7% to 34.5%. Interestingly, dual emission peaks are observed at low temperatures due to the presence of two excitation structures on the potential energy surface of the triplet state excited states. The self-trapped excitons of Cu:(PEA)2ZnBr4 can cross this potential barrier between the two triplet-state excited states by acquiring thermal energy, but Cu:(PEA)2ZnCl4 cannot overcome the barrier due to the different molecular environments. For the first time, using Cu:(PEA)2ZnX4 as the light-emitting layers, sky-blue LEDs with an external quantum efficiency (EQE) of 0.45% and 0.83% for Cu:(PEA)2ZnCl4 and Cu:(PEA)2ZnBr4 were obtained, demonstrating that broadband light-emitting lead-free metal halides have great potential for sky-blue light-emitting diode applications.


 Please wait while we load your content...
Please wait while we load your content...