A method for identifying the cause of inefficient salt-doping in organic semiconductors†
Abstract
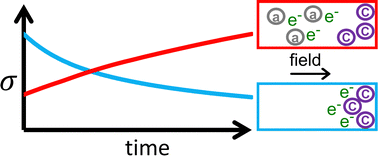
Doping to enhance the electrical conductivity of organic semiconductors is not without its challenges: The efficacy of this process depends on many factors and it is not always clear how to remedy poor doping. In the case of doping with salts, one of the possible causes of poor doping is a limited yield of integer charge transfer resulting in the presence of both cations and anions in the film. The charge of such ions can severely limit the electrical conductivity, but their presence is not easily determined. Here we introduce a set of simple conductivity measurements to determine whether poor doping in the case where the dopant is a salt is due to limited integer charge transfer. By tracking how the conductivity changes over time when applying a bias voltage for an extended amount of time we can pinpoint whether unwanted ions are present in the film. Firstly, we introduce the principle of this approach by performing numerical simulations that include the movement of ions. We show that the conductivity can increase or decrease depending on the type of ions present in the film. Next, we show that the movement of these dopant ions causes a build-up of space-charge, which makes the current–voltage characteristic non-linear. Next, we illustrate how this approach may be used in practice by doping a fullerene derivative with a series of organic salts. We thus provide a tool to make the optimization of doping more rational.
- This article is part of the themed collection: Special issue in honour of Kees Hummelen


 Please wait while we load your content...
Please wait while we load your content...