Fast charge transfer between iodide ions and a delocalized electron system on the graphite surface for boosting hydrogen production†
Abstract
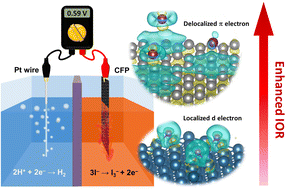
Developing new catalysts to reduce the energy barrier for hydrogen production from water splitting has been an important research topic. It is well-known that the water-splitting voltage is limited by a theoretical potential of 1.23 V of the oxygen evolution reaction (OER). Recently, a new electrolytic method using the iodide oxidation reaction (IOR) instead of the OER has been proposed to produce the valuable chemicals iodine and hydrogen efficiently with a reduced operating voltage. In this work, we found that the graphite-based materials with a delocalized π electron system exhibited fast charge transfer with physically adsorbed iodide ions, thus revealing desirable IOR catalytic activity. The carbon fiber paper with a graphite structure was chosen as the catalyst and exhibited an onset potential of 0.54 V (vs. RHE) for the IOR, which is close to the standard potential of iodide oxidation. Also, the CFP demonstrated a minimum Tafel slope of 47.78 mV dec−1 in the electrochemical reaction with significant stability at a current density of 10 mA cm−2 for over 18 hours. Moreover, in a two-electrode system, a cell voltage of only 0.59 V was required to provide a current density of 10 mA cm−2 for the IOR and hydrogen evolution reaction (HER). In terms of energy costs, our HER and IOR system can reduce energy consumption by 65% compared to conventional OER-based water electrolysis. The overall results indicate that our findings provide a new route to facilitate the industrial application of hydrogen production via electrochemical reactions.


 Please wait while we load your content...
Please wait while we load your content...