CO2-to-CH4 electroreduction over scalable Cu-porphyrin based organic polymers promoted by direct auxiliary bonding interaction†
Abstract
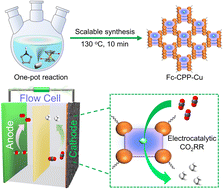
The scale-up syntheses of electrocatalysts with rationally designed functional promoter groups that can directly interact with catalytic-centres are much desired for CO2 electroreduction. Here, we propose an in situ copolymerization method that can be applied for the scale-up syntheses of metalloporphyrin based conjugated porous polymers with directly interacting catalytic centres and promoter groups. The thus-obtained materials possess the characteristics of high porosity, adjustable morphology, abundant catalytic sites and directly interacting promoter groups. In particular, CPP–Cu with ferrocene as the promoter group exhibits a FECH4+C2H4 of 94.0% (FECH4, 75.9%, FEC2H4, 18.1%, −0.9 V), which is much higher than that of Cu-porphyrin, Bz-CPP-Cu and the physical-mixture and represents one of the best electro-catalysts so far. DFT calculations reveal that the directly-interacting ferrocene groups could enhance the electron-cloud density of Cu-porphyrin and possess strong adsorption-ability to OH* to kinetically improve the proton-coupled electron transfer for the preferential CO2-to-CH4 pathway. Noteworthily, the synthesis-strategy is easy to scale-up (∼10 g in a batch-experiment) and the reaction-time can be as little as 10 min under microwave-conditions.


 Please wait while we load your content...
Please wait while we load your content...