Scanning transmission X-ray microscopy studies of electrochemical activation and capacitive behavior of Mn3O4 supercapacitor electrodes†
Abstract
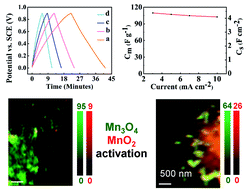
This investigation, which is motivated by promising pseudocapacitive properties of Mn3O4 for energy storage in cathodes of supercapacitors, addresses the need to understand both the activation and the charge storage mechanisms of Mn3O4 electrodes. Specific activation protocols are shown to result in significant capacitance increase during cycling. For the first time scanning transmission X-ray microscopy (STXM) is used for analysis of Mn3O4 activation. STXM analyses at the Mn 2p and O 1s edges provide chemical mapping of different oxidation states with high spatial resolution. Mn3O4-carbon nanotube composite electrodes with commercially important high active mass loading of 40 mg cm−2 are prepared using quercetin dispersant. The catecholate type polyaromatic quercetin facilitates co-dispersion of Mn3O4 with carbon nanotubes and allows enhanced electrode performance at high active mass loadings. Cyclic voltammetry, electrochemical impedance spectroscopy and galvanostatic charge–discharge are used for the capacitance monitoring during electrode activation. Two strategies are used for electrode activation in Na2SO4 electrolyte: electrochemical cycling at different scan rates and continuous cycling at a fixed scan rate. The capacitance variations are linked to STXM observations, which show gradual oxidation of Mn3O4 to form MnO2 on the particle surface and in the bulk. The pseudocapacitive charge storage mechanism involves in situ oxidation of Mn3O4 and redox reactions of Mn4+/Mn3+ species on the particle surface and in the bulk.


 Please wait while we load your content...
Please wait while we load your content...