The proton uptake process in double perovskite triple ionic-electronic conducting oxides for protonic ceramic cells†
Abstract
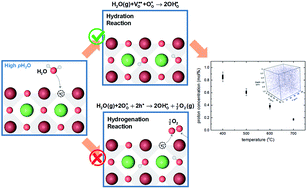
Triple ionic-electronic conducting oxides, in which electron holes, oxygen ions, and protons are simultaneously mobile, are garnering massive attention as potential oxygen electrodes in protonic ceramic cells. While their high electrocatalytic activity and proton transport ability generally provide opportunities for enhanced performance of electrochemical cells, the lack of fundamental knowledge on the proton uptake mechanism hinders the rational modification and design of materials. Herein, we investigate the proton uptake mechanism in double-perovskite PrBa0.5Sr0.5Co1.5Fe0.5O5+δ (PBSCF), a widely used triple ionic-electronic conducting oxide. The proton solubility of PBSCF is examined by direct observation of protons using secondary ion mass spectrometry. We then comprehensively discuss the correlations between previously hypothesized proton uptake mechanisms (hydration vs. hydrogenation) and experimentally measured properties such as mass and conductivity changes. This work provides a guideline for establishing the design principles of triple ionic-electronic conducting oxides based on mechanistic understanding.


 Please wait while we load your content...
Please wait while we load your content...