Low-temperature liquid reflux synthesis of core@shell structured Ni@Fe-doped NiCo nanoparticles decorated on carbon nanotubes as a bifunctional electrocatalyst for Zn–air batteries†
Abstract
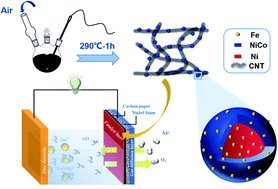
Exploring high-efficiency electrocatalysts for the oxygen reduction reaction (ORR) and oxygen evolution reaction (OER) was a decisive parameter for metal–air batteries, fuel batteries, and water splitting. In this work, core@shell structured Ni@Fe-doped NiCo nanoparticles physically decorated on carbon nanotubes (Fe/Ni@NiCo-CNT) were synthesized through a facile low-temperature liquid reflux strategy. Electron microscope characterization evidenced a unique core@shell structure composed of a metallic Ni core and NiCo alloy shell with Fe element doping. Electrochemical measurements illustrated that the hybrid Fe/Ni@NiCo-CNT electrocatalyst showed superior ORR and OER activities, approaching and even exceeding those of commercial Pt/C and RuO2 catalysts. The about 4 nm-thick shell of the Fe-doped NiCo alloy combined with the synergetic effects between nanoparticles and carbon nanotubes (CNTs) made dominant contributions to increasing the electrochemically active surface area and accelerating electrocatalytic kinetics. When Fe/Ni@NiCo-CNT was applied in Zn–air batteries (ZABs), the primary ZABs delivered an open circuit potential of 1.427 V and a specific capacity of 796.5 mA h gZn−1. Moreover, the rechargeable ZABs surprisingly sustained for more than 1500 cycles with steady voltage polarization at 5 mA cm−2. This cost-effective strategy shows important guidance for the structural engineering of other metallic alloy materials and further sheds light on developing efficient bifunctional oxygen catalysts for renewable energy conversion devices.


 Please wait while we load your content...
Please wait while we load your content...