Redox-driven strong interfacial interactions between MnO2 and covalent organic nanosheets for efficient oxygen reduction electrocatalysis†
Abstract
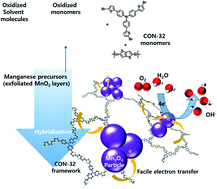
Hybridization between inorganic and organic nanostructures has attracted a great deal of research interest because of its usefulness as an efficient synthetic method for high-performance electrocatalysts. In this study, a novel synthetic strategy to explore efficient electrocatalysts was developed based on a redox-driven strong electronic coupling between MnO2 and covalent organic nanosheets (CONs), denoted as CON-32. This synthetic strategy is based on the formation of robust interfacial bonds, such as Mn–O–R and Mn–O–S, which are generated during the redox reaction between MnO2 and CON-32. Heat treatment of the as-prepared hybrids at elevated temperatures led to the carbonization of CON-32 and thus the improvement of electrical conductivity. Both the as-prepared and carbonized hybrids delivered significantly improved electrocatalytic properties for the oxygen reduction reaction (ORR) compared to the precursors COF, MnO2, and Mn3O4, with a higher half-wave potential and a larger electron transfer number being achieved; these results highlight the beneficial effects of hybridization and carbonization. The high efficacy of hybridization in improving the electrocatalyst functionality can be ascribed to the remarkable enhancement of the ORR kinetics and the interfacial electron transfer to the hydroxide ions, which is caused by the synergetic combination of conductive CONs and the electrocatalytically active MnO2, as confirmed by in situ electron paramagnetic resonance spectroscopy.


 Please wait while we load your content...
Please wait while we load your content...