Facile fabrication of functionalized pyrimidine derivatives: constructing a new family of high performance and less sensitive energetic compounds†‡
Abstract
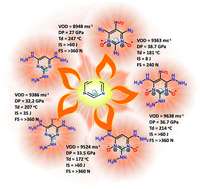
A series of new pyrimidine-based nitrogen-rich green energetic materials were effectively synthesized from commercially available inexpensive starting materials, with short reaction times, easy isolation, and essentially quantitative yields. All the molecules were fully characterized using IR, multinuclear nuclear magnetic resonance (NMR), and mass spectra. Compounds 2, 4 and 5 were characterized by 15N NMR spectroscopy, and compound 4 was confirmed by single-crystal X-ray diffraction studies. The physicochemical properties of the pyrimidine derivatives were investigated theoretically and experimentally in detail. Compounds 2, 3, 9, 10 and 12 exhibit excellent detonation properties, good thermostability and low sensitivity comparable with secondary benchmark explosives RDX and HMX. It is noteworthy that for compounds 3, 9 and 10, the values of the heats of formation, detonation velocities and pressures, decomposition temperatures, and impact sensitivities were determined to be >250 kJ mol−1, >9380 m s−1, >32 GPa, >170 °C, and >30 J, respectively. In addition, the activation energy and nonisothermal thermokinetic parameters for compounds 2, 9 and 10 were determined by using the Kissinger method. It was found that incorporating hydrazine, nitro, and N-oxide groups into the pyrimidine backbone leads to strong non-bonded interactions, which helps to enhance the thermal stability and lower the sensitivity. These results are expected to assist in improving the synthesis strategy of balancing the detonation performance and safety parameters of a new generation of green energetic materials.


 Please wait while we load your content...
Please wait while we load your content...