Room-temperature all-solid-state lithium metal batteries based on ultrathin polymeric electrolytes†
Abstract
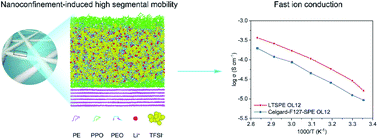
Flexible all solid-state batteries that combine polymer electrolytes and lithium metal are required for powering wearable electronic devices as they embody high flexibility, safety and energy density. However, it has been challenging to develop room temperature flexible solid-state batteries because of the low ionic conductivities of polymeric ion conductors. Here, we report a highly conductive polymer electrolyte based on lowly viscous polyethylene oxide ion conductors confined inside an ultrastrong and ultrathin nanofibrous polyethylene membrane. The newly developed solid polymer electrolyte exhibits high room temperature charge transfer conductance (0.07 S) and excellent compatibility with Li metal (cycling time > 3500 hours at 0.1 mA cm−2) at 30 °C. Large-scale molecular dynamics simulations show that the high ionic conductivity is because of the enhanced segmental mobility of confined PEO inside the nanomembrane and the strong charge–dipole interactions between the PEO and lithium ions. This work provides a viable solution toward room temperature workable solid-state batteries that meet the requirements for wearable electronic devices.


 Please wait while we load your content...
Please wait while we load your content...