Improving oxygen evolution activity by constructing a perylene imide based Z-scheme heterojunction†
Abstract
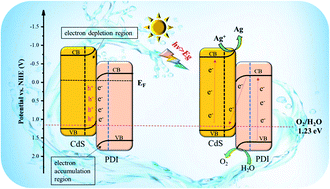
Solar water splitting is an eco-friendly technology to produce clean energy, but the water oxidation half reaction hinders the overall water splitting due to four electron transfer processes. A Z-scheme photocatalytic system could effectively promote the separation of photogenerated carriers and keep strong redox ability. However, to date, research on Z-scheme heterojunction materials for the photocatalytic oxygen evolution half reaction has been very limited, and the mechanism of enhancing oxygen evolution activity has never been studied in detail. In this work, for the first time a CdS/PDI Z-scheme heterojunction system with high activity for O2 evolution was constructed. The electron transfer pathway and photoinduced carrier separation mechanism were deeply studied. Based on experimental and calculation results, the Fermi energy level of CdS was higher than that of the PDI polymer and a strong built-in electric field was formed. The direction of the built-in electric field is from CdS to PDI. Meanwhile under visible light illumination, the photogenerated electrons transferred along the Z-scheme transfer pathway, which not only significantly increased the separation efficiency of electrons/holes but also retained the stronger oxidability of holes in PDI, thus dramatically enhancing the oxygen evolution activity of PDI. The highest O2 evolution rate on 15%CdS/PDI was 1392.29 μmol g−1 h−1, and it was 2.0 times higher than that of pristine PDI under visible light illumination. Theoretical calculation results indicated that the formation of the intermediate state of OOH* was the rate determining step for PDI in the photocatalytic water oxidation reaction, and the CdS/PDI Z-scheme heterojunction could greatly reduce the energy barrier of the rate determining step, which is also a crucial factor for the enhancement of the photocatalytic oxygen evolution activity of the CdS/PDI Z-scheme heterostructure.


 Please wait while we load your content...
Please wait while we load your content...