A three-dimensional ordered honeycomb nanostructure anchored with Pt–N active sites via self-assembly of a block copolymer: an efficient electrocatalyst towards the oxygen reduction reaction in fuel cells†
Abstract
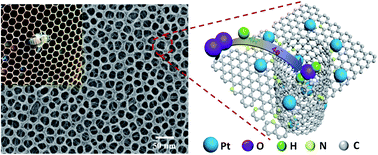
Mesoporous Pt-containing nanocomposites with well-organized pores are desirable for fuel cells as well as sensors, electronics, and various chemical reactions. However, it remains challenging to construct three-dimensional (3D) ordered honeycomb-like (OHC) nanostructures with Pt species anchored in the mesopores. Herein, we show for the first time an in situ strategy of developing an N-doped ordered honeycomb (N-OHC) nanopattern with well-dispersed Pt–N2 moieties by using the self-assembly of a block copolymer (BCP). The as-mentioned Pt including Pt single atoms (SAs) and Pt ∼2.5 nm ultrafine nanoparticles (NPs) was hierarchically located on the inner walls and the outer surfaces of the N-OHC mesopores (Pt/N-OHC), forming well-dispersed Pt–N active sites and showing efficient catalytic activity towards the oxygen reduction reaction (ORR). By changing the film thickness of the pristine Pt/BCP template, double-layered Pt/N-OHC could be designed and the ORR activity could be correspondingly improved, for which a current density of 1.60 A cm−2@0.6 V and a peak power density of 1.07 W cm−2 were observed at a very low Pt-loading of 0.04 mg cm−2, better than 1.21 A cm−2@0.6 V and 0.79 W cm−2 of the commercial Pt/C catalyst at a Pt-loading of 0.15 mg cm−2. The electron donating behavior of Pt–N2 and the pathway for O2 reduction were investigated via the density functional theory (DFT) computation. The electron transfer from Pt to N gave rise to the formation of Pt–N covalent bonds, which resulted in a lower d band center and a weaker O adsorption energy and endowed the Pt/N-OHCs with enhanced ORR activity.


 Please wait while we load your content...
Please wait while we load your content...